
Since we were asked for the moles of silver chromate that would disolve in 1.00 L, the final answer is: Since there is a 2:1 ratio between the moles of aqueous silver ion and the moles of silver chromate that dissolved, 1.5 x 10 -5 M is the molar solubility of Ag 2CrO 4 in 0.010 M K 2CrO 4 solution. 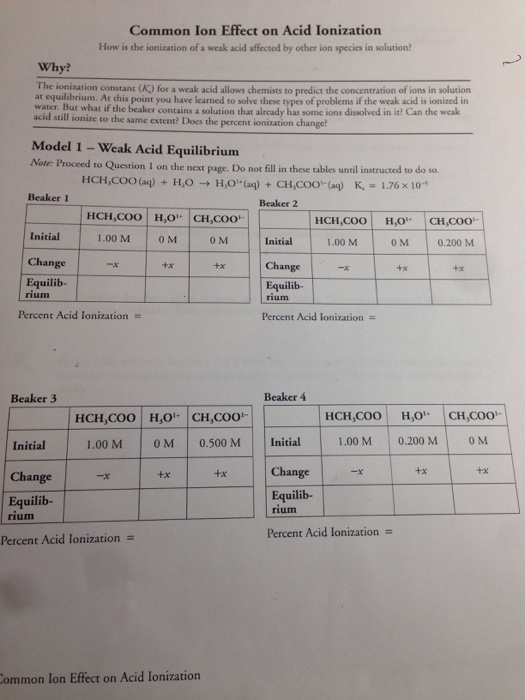
K sp for Ag 2CrO 4 = 9.0 x 10 -12.ġ) Concentration of dichromate ion from potassium chromate: Problem #3: Calculate the number of moles of Ag 2CrO 4 that will dissolve in 1.00 L of 0.010 M K 2CrO 4 solution. Note how zinc hydroxide would precipitate even when the solution is slightly acidic. X = 5.91657 x 10 -8 M (I kept a few guard digits.) 
Problem #2: Calculate the pH at which zinc hydroxide just starts to precipitate from a 0.00857 M solution of zinc nitrate. Substituting into the K sp expression:Īny sodium hydroxide solution greater than 0.33 M will reduce the to less than 1.1 x 10¯ 10 M. What minimum OH¯ concentration must be attained (for example, by adding NaOH) to decrease the Mg 2+ concentration in a solution of Mg(NO 3) 2 to less than 1.1 x 10¯ 10 M? Problem #1: The solubility product of Mg(OH) 2 is 1.2 x 10¯ 11. The Common Ion Effect - Problems 1 - 10 The Common Ion Effect
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
